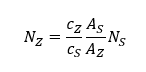Metalloprotein Analysis Template
Metallo Protein Analysis
Metallo-protein analysis focuses on studying proteins that contain metal ions as part of their structure. It identifies the type and amount of metal present and examines how the metal interacts with the protein. Techniques like UV–Vis spectroscopy, ICP-MS, X-ray crystallography, and EPR help characterize metal centers. This analysis reveals the metal’s role in catalysis, electron transfer, stability, and protein activity.
It also helps understand oxidation states, metal-binding sites, and coordination geometry. Metallo-protein analysis is crucial because nearly one-third of all proteins depend on metals. It aids in identifying how disruptions in metal binding can lead to diseases.
Ion Beam Analysis of Metals in Proteins
Geoffrey W. Grime


📰
Geoffrey W. Grime et al. Journal of the American Chemical Society 2020 142 (1), 185-197. DOI: 10.1021/jacs.9b09186
Why measure metals in proteins?
- Proteins are basic components of biochemical processes including cell function, tissue building, disease processes and metal transport (e.g. haemoglobin).
- Proteins are linear chains of amino acids folded into complex 3D shapes. Shape determines function.
- Many proteins contain small numbers of metal atoms. X-ray crystallography is routinely used to determine the position of each non-hydrogen atom in a protein, including metals. However, this relies on fitting the electron density, which does not allow the unambiguous identification of the metal.
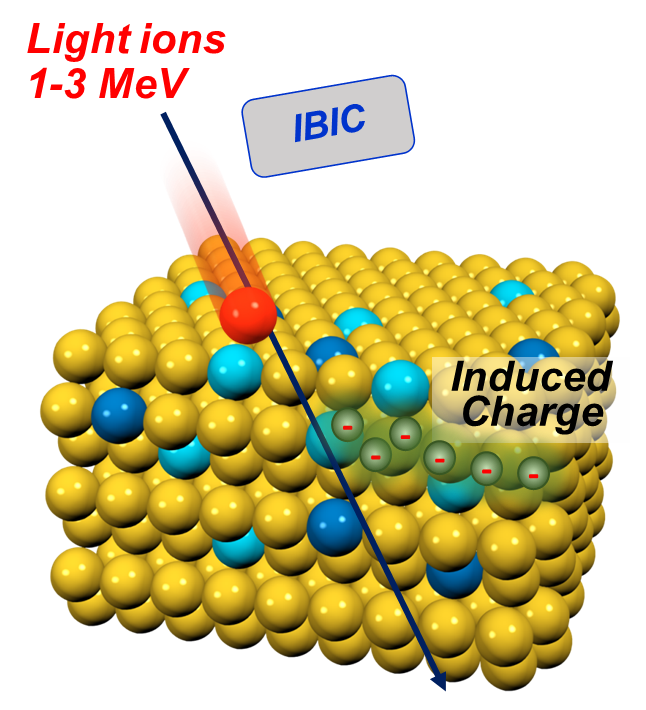
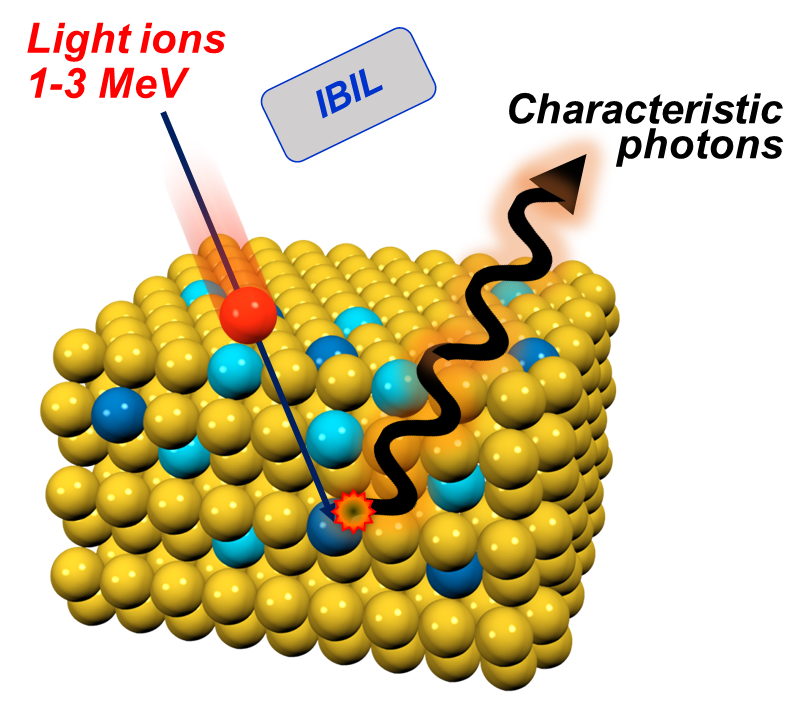
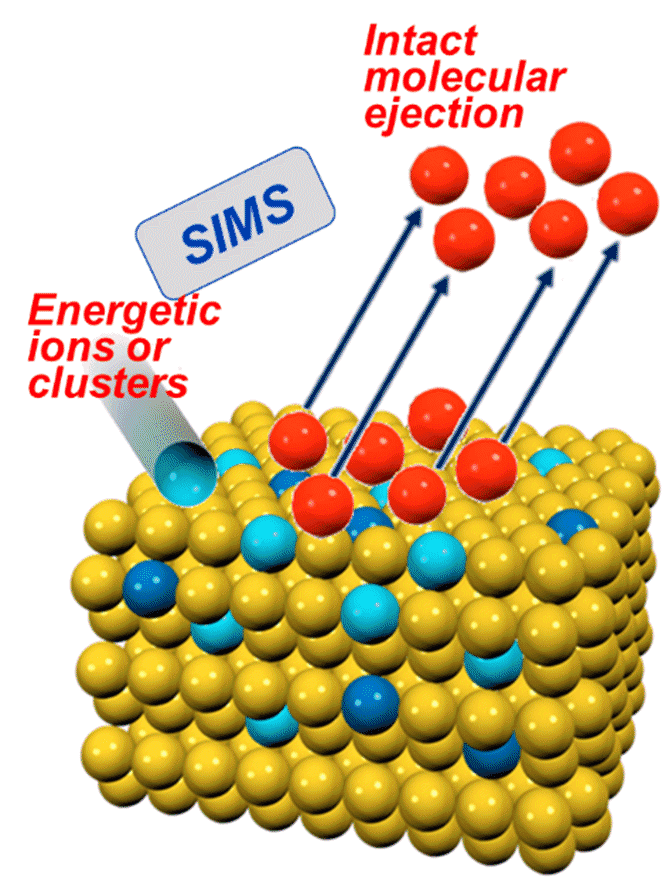
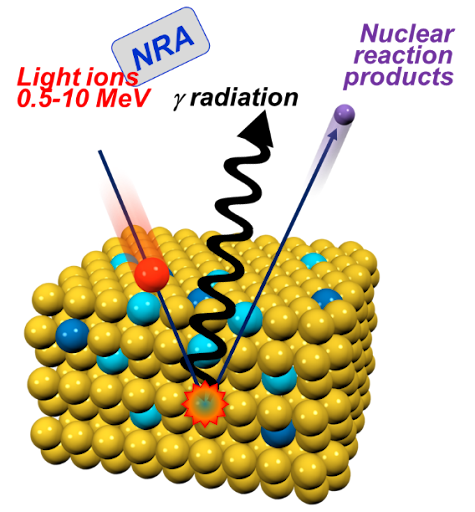
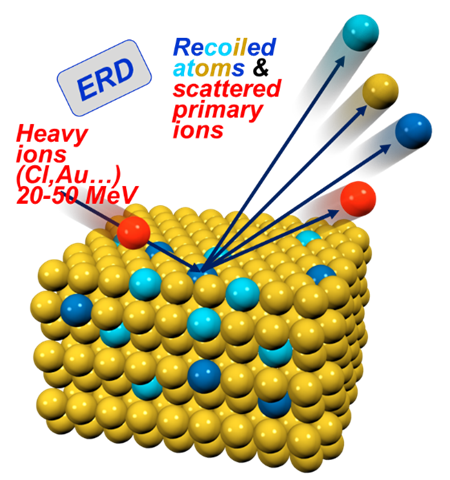
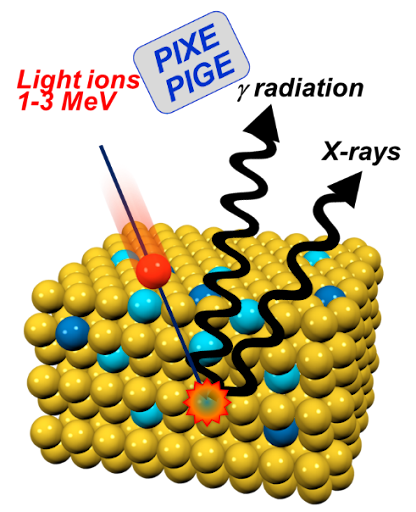
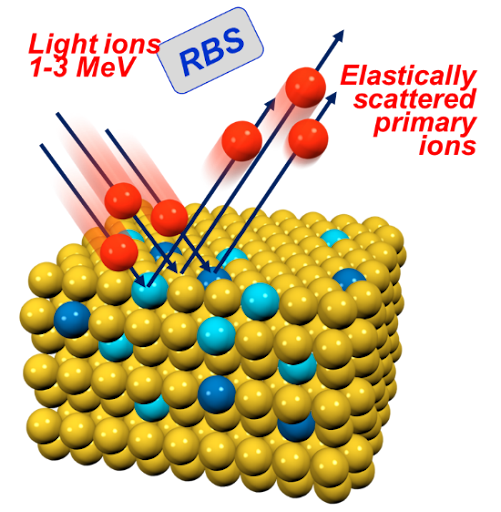
- Micro particle induced X-ray emission (µPIXE) can identify and quantify unknown metal atoms in proteins.
The scale of the problem
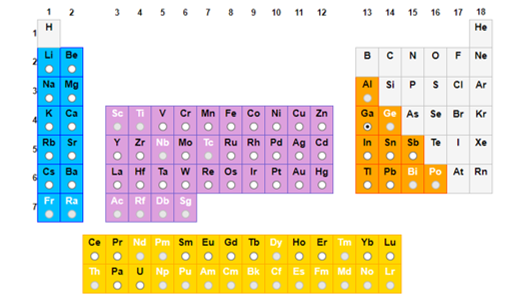
☐ Over one third of all proteins with known 3-D structure are identified as containing metal atoms or binding sites.
☐ Elements with round white symbols have been identified in a protein structure.
Our work indicates that up to 30% of metal assignments may be incorrect, possibly leading to erroneous biological conclusions being drawn from the structures.
Ion Beam Analysis of Metals in Proteins
Geoffrey W. Grime


Analysis of metals in proteins using PIXE
☐ Absolute measurement of metal concentration is not easy
☐ Metal concentrations are low
☐ Proteins must be dissolved in buffer solutions so amount of protein may not be well known
The scale of the problem
Elspeth’s insight in 1993:
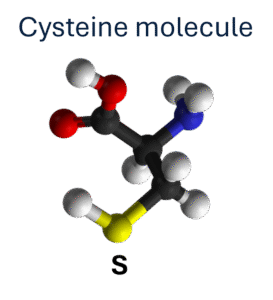
Most proteins have known numbers of the amino acids methionine and cysteine in their sequence with each contain one sulphur atom. Use the known number of S atoms as an internal standard!
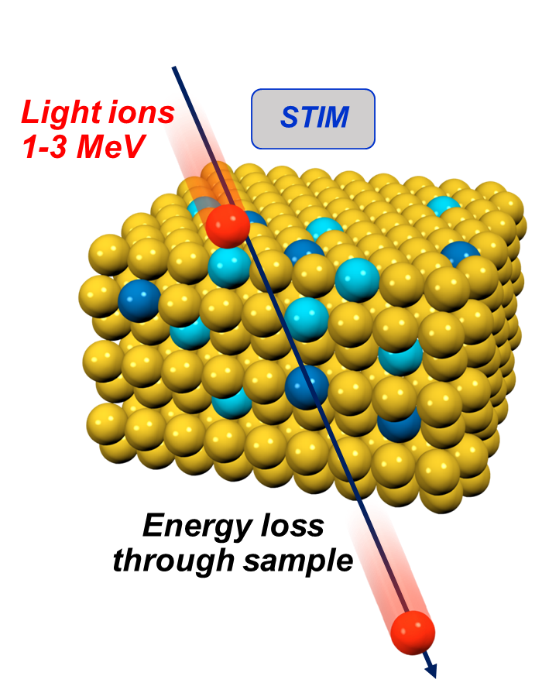
c is measured concentration
A is atomic weight
N is the number of detected metal atoms of interest per molecule
Z, S refer to the metal atom of interest and sulphur
☐ Using ratios eliminates many sources of error affecting absolute measurement.
☐ Other elements may be used as the reference (e.g. P in nucleic acids, Se following seleno-methionine substitution.)
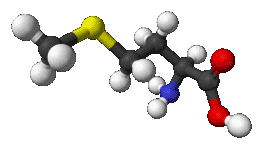
Methionine molecule
Ion Beam Analysis of Metals in Proteins
Geoffrey W. Grime


📰
Pounot, Kevin et al. Biophysical Journal, 120, Issue 5, 886 – 898
DOI: 10.1016/j.bpj.2020.11.2280
Human Insulin: The importance of Zinc (Zn)
Diabetics who inject insulin may suffer from amyloidosis, an inflammation at the injection site.
- Caused by insulin aggregating to form fibrils and spherulites that compromise its action.
- PIXE showed that zinc is expelled on aggregation of insulin.
- PIXE was crucial for the study and this finding is helping develop treatments for amyloidosis.